CAR-T Therapy in China for Relapsed Multiple Myeloma: One International Patient’s Journey to Beijing
After standard treatment options were exhausted at home, a 45-year-old international patient traveled to Beijing to be evaluated for CAR-T therapy for relapsed and refractory multiple myeloma. His case highlights not only the medical decision-making behind cross-border cancer care, but also the coordination that helped his family navigate treatment in an unfamiliar healthcare system.
In early December, a patient we will call Alex began reaching out from overseas. Over the next three weeks, his family exchanged dozens of emails with a medical coordination team in Beijing while exploring whether treatment in China might still be possible.
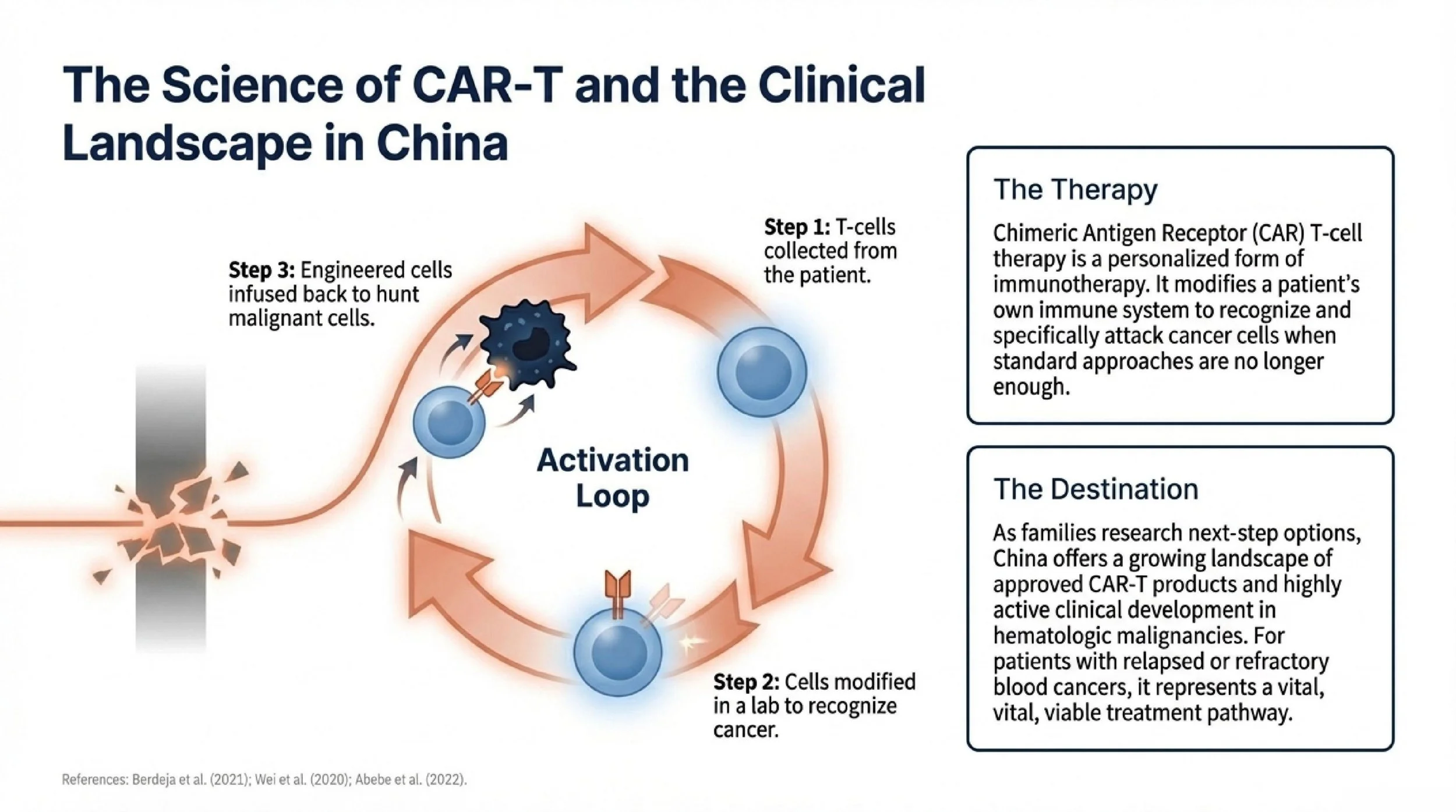
Alex, 45, had relapsed and refractory multiple myeloma. He had already undergone multiple lines of treatment in his home country, including standard first- and second-line chemotherapy. He had achieved remission twice, but by November, the disease had returned again. According to his local care team, there were no clear conventional options left to pursue.
At that point, Alex and his family began looking beyond their home country.
Why Alex Began Looking at CAR-T Therapy in China
As they researched next-step options, Alex’s family came across China’s growing experience in CAR-T therapy, including approved products and active clinical development in hematologic malignancies. For patients with relapsed or refractory blood cancers, CAR-T therapy has become an important treatment pathway in selected cases, particularly after prior therapies have failed.¹ ² ³
CAR-T, or chimeric antigen receptor T-cell therapy, is a personalized form of immunotherapy. A patient’s own T cells are collected and modified to recognize and attack cancer cells. For some patients with relapsed or refractory disease, it may offer another treatment option when standard approaches are no longer enough.
Still, deciding to seek treatment abroad was not simple.
The Real Barriers to Cross-Border Cancer Care
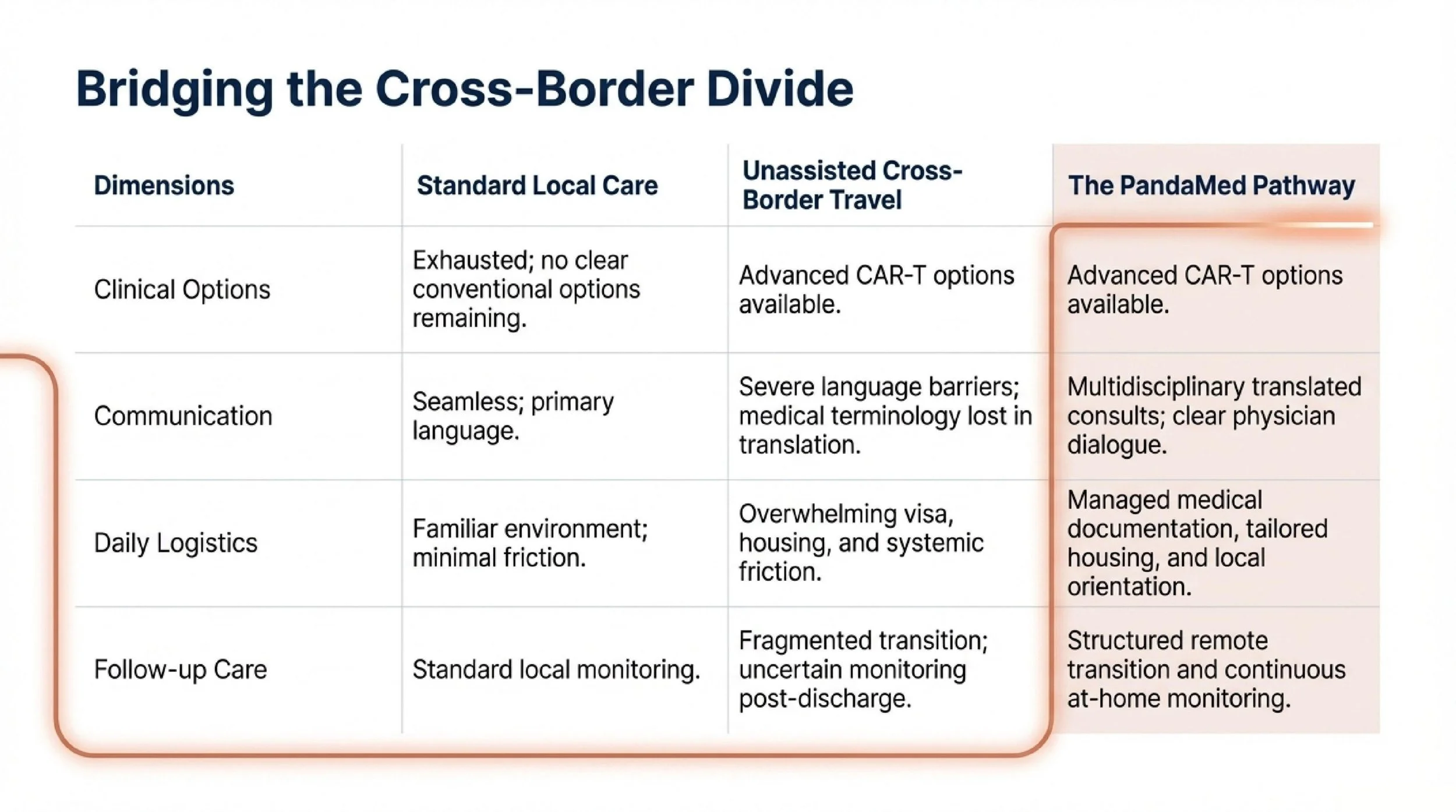
For Alex and his family, the challenge was not only medical. They had never pursued treatment in another country before. They were worried about how to evaluate hospitals from afar, how to communicate across language differences, how treatment would be explained, and how daily life would work during a long stay overseas.
Those concerns are common among international patients. Even when a treatment option looks promising on paper, uncertainty about process and logistics can delay decision-making.
To help the family make an informed choice, a Beijing-based team arranged a remote multidisciplinary consultation. Before discussing treatment details, the family was introduced to the clinical setting and care process so they could better understand what treatment in Beijing would actually involve. A hematology specialist then reviewed the proposed CAR-T pathway in practical terms, including candidacy, treatment steps, potential side effects, expected timeline, and how Alex’s prior treatment history might affect planning.
That conversation did not remove all uncertainty, but it gave the family something they had not had before: a clearer basis for decision-making.
When visa-related issues later threatened to delay travel, the coordination team assisted with the medical documentation needed to support the application process so the family could move forward without losing more time.
Arrival in Beijing and a New Set of Questions
Once the family arrived in Beijing, the first priority was not treatment itself, but confirmation.
Alex was admitted for evaluation and underwent the standard workup needed before CAR-T planning could proceed, including blood testing, bone marrow assessment, and imaging. Based on those findings, the clinical team considered him an appropriate candidate to move forward.
Then came an unexpected complication.
During pre-treatment screening, Alex tested positive for cytomegalovirus, or CMV.⁴ In patients preparing for CAR-T therapy, this matters. CMV reactivation can become clinically significant during periods of immune suppression and may increase treatment-related risk if not addressed appropriately.⁵
Instead of proceeding immediately, the medical team adjusted course and initiated antiviral management first. For the family, this waiting period was difficult. After traveling internationally for treatment, any delay can feel overwhelming. But from a clinical standpoint, this step was necessary. Roughly two weeks later, Alex’s CMV markers turned negative, and treatment planning was able to continue.
Treatment Required More Than a Procedure
Alex then moved through the key stages of CAR-T treatment, including cell collection, preparative therapy, and CAR-T cell infusion, with close monitoring throughout.
Because CAR-T therapy can involve serious short-term risks, careful observation is an essential part of care. Alex was followed by a multidisciplinary team during the most critical periods after treatment, with attention to both response and potential complications.
At the same time, the family was also trying to adapt to daily life in a new country. To reduce unnecessary stress during a demanding treatment period, practical support was arranged around communication, local orientation, and longer-stay living needs. For example, housing was selected with the family’s day-to-day routines in mind, including access to cooking facilities during recovery. These details did not replace medical care, but they helped make a prolonged cross-border treatment period more manageable.
That distinction matters. In international care, good outcomes depend not only on treatment decisions, but also on whether the patient and family can realistically sustain the process.
A Positive Early Outcome After a Difficult Course
After nearly two months in Beijing, Alex’s disease burden was reported to be under control, and his overall physical condition improved steadily during recovery. At discharge, he spoke positively about both the clinical rigor and the consistency of support he had received during treatment.
His care did not end when he left China. Before returning home, a remote follow-up plan was established so his progress could continue to be monitored after discharge. Ongoing guidance included medication management, rehabilitation support, and practical recommendations related to diet and physical recovery.
For international patients, that transition phase is often one of the biggest concerns. Having a follow-up framework in place can make the return home feel more structured and less uncertain.
What This Case May Mean for Other International Patients
Alex’s case reflects one patient’s individual experience. Treatment suitability and outcomes vary based on diagnosis, disease status, prior treatment history, physician evaluation, and treatment response.
Still, his experience illustrates several important points for international patients exploring cancer treatment in China: access to treatment begins with careful case review, cross-border care requires both medical and logistical planning, and unexpected issues may need to be addressed before treatment can proceed safely.
For families facing relapsed or refractory disease, clarity is often the first need. Before deciding whether travel is appropriate, patients usually need help understanding candidacy, medical records, treatment timing, and what the process may actually involve.
If you or a family member are exploring whether CAR-T therapy or other cancer treatment in China may be an option, you can contact the PandaMed Global team at contact@pandamedglobal.com. We can help you organize medical records, support cross-border communication, and understand the next steps for physician review and treatment planning.
References
Berdeja, J. G., et al. (2021). Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study. The Lancet, 398(10297), 314-324.
Wei, J., et al. (2020). The model of comprehensive evaluation of CAR-T cell therapy in China. Cancer Cell International, 20(1), 1-10.
Abebe, E. C., et al. (2022). Ciltacabtagene autoleucel: The second anti-BCMA CAR T-cell therapeutic armamentarium of relapsed or refractory multiple myeloma. Frontiers in Immunology.
Kampouri, E., et al. (2024). Incidence and outcomes of cytomegalovirus reactivation after chimeric antigen receptor T-cell therapy. Blood Advances, 8(14), 3813.
Zhang, M., et al. (2026). Cytomegalovirus Infection and Clinical Outcomes in Hospitalized Chimeric Antigen Receptor T-Cell Therapy Recipients. Frontiers in Immunology / PMC.