Refractory SLE-ITP Failed Every Drug — How CD19 CAR-T in Beijing Achieved Complete Remission
If you or a family member has been living with lupus-related immune thrombocytopenia that hasn't responded to steroids, rituximab, TPO agonists, or any combination of standard immunosuppressants, you already know the reality: each failed therapy narrows the next option and raises the bleeding risk. Platelet counts stuck below 20×10⁹/L mean every week carries the possibility of a catastrophic hemorrhage.
What most patients outside China don't yet know is that Beijing's top rheumatology and hematology centers have moved beyond salvage immunosuppression entirely. In July 2024, the New England Journal of Medicine published the world's first reported case of CD19-targeted CAR-T cell therapy for SLE-ITP — performed at Peking Union Medical College Hospital. The patient's platelets recovered from 4×10⁹/L to 109×10⁹/L within six months, with complete remission and discontinuation of all immunosuppressants.
This article explains the clinical pathway, the hospitals leading this work, what records you need, and how international patients can realistically access these programs.
Quick Decision Snapshot
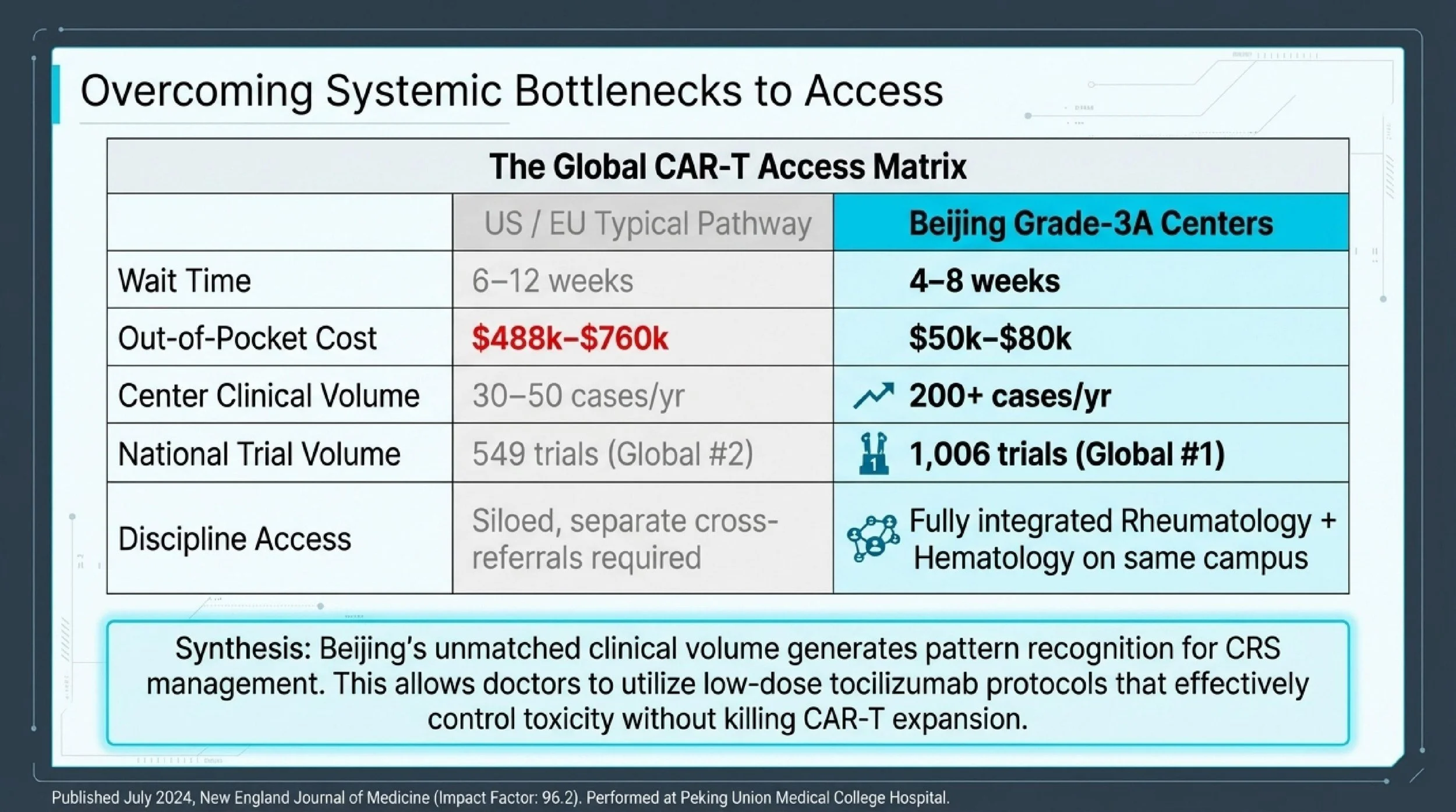
| Decision Factor | US / EU Typical Pathway | Beijing Grade-3A Public Hospitals |
|---|---|---|
| CAR-T Scheduling Wait | 6–12 weeks | 4–8 weeks |
| Annual CAR-T Clinical Volume per Center | 30–50 cases | 200+ cases |
| Out-of-Pocket Cost (Commercial CAR-T) | $488,000–$760,000 | $50,000–$80,000 |
| National CAR-T Clinical Trial Volume | 549 registered trials (Global #2) | 1,006 registered trials (Global #1) |
| Dual-Discipline Access (Rheumatology + Hematology) | Separate referrals; often different institutions | Integrated within same hospital campus |
Sources: 2026 Cost Analysis of Sylk Health; ClinicalTrials.gov
If you have existing medical records — lab work, rheumatology reports, bone marrow biopsy, or treatment history — you can send them to contact@pandamedglobal.com to check whether they're enough for an initial records review and to ask what to prepare next.
The Case That Changed the Field: World's First CAR-T for SLE-ITP
The landmark patient had been diagnosed with SLE in 2014. For nine years, severe thrombocytopenia persisted below 20×10⁹/L despite an exhaustive list of failed therapies: methylprednisolone pulse, hydroxychloroquine, IVIG, tacrolimus, cyclosporine, sirolimus, rituximab, danazol, eltrombopag, and TPO receptor agonists.
By the time the team at Peking Union Medical College Hospital intervened, the platelet count had dropped to 4×10⁹/L — less than 10% of the normal lower limit. The risk of spontaneous intracranial or gastrointestinal hemorrhage was immediate and ongoing.
The treatment was autologous CD19-targeted CAR-T cell therapy: the patient's own T-cells were collected, genetically engineered to express chimeric antigen receptors against CD19-positive B-cells, and reinfused. The goal was to eliminate the pathogenic B-cell population responsible for producing the autoantibodies driving both SLE activity and platelet destruction.
Results published in the New England Journal of Medicine (IF: 96.2), July 2024:
The platelet count recovered from 4×10⁹/L to 109×10⁹/L within six months. The patient achieved clinical complete remission. Complete CD19+ B-cell clearance was confirmed by Day 28, with B-cell reconstitution beginning by Month 3. ANA titers dropped significantly. All immunosuppressants and corticosteroids were successfully discontinued. No severe cytokine release syndrome (CRS) or immune effector cell-associated neurotoxicity syndrome (ICANS) was observed.
This was not an incremental improvement. It was a proof-of-concept that CAR-T therapy — originally developed for hematologic malignancies — could achieve durable remission in refractory autoimmune cytopenias where every conventional agent had failed.
Source: New England Journal of Medicine (2024); Peking Union Medical College Hospital Official Report
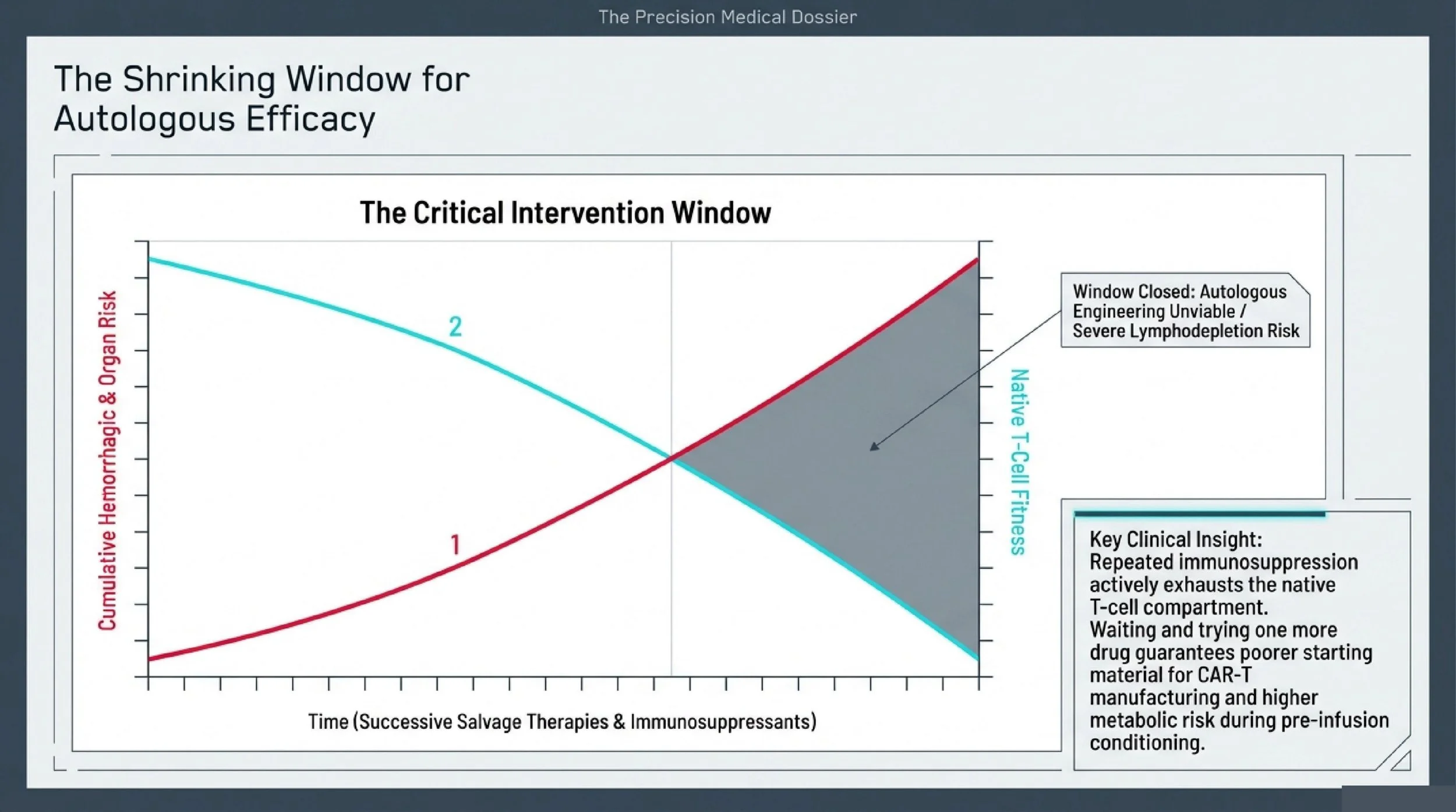
Why Delay Is Dangerous in Refractory SLE-ITP
For patients whose platelet counts remain critically low despite multiple lines of therapy, the instinct to "wait and see" or "try one more drug" carries compounding risks that directly undermine the chance of a successful CAR-T outcome later.
Hemorrhagic risk accumulates, not stabilizes. Persistent severe thrombocytopenia below 20×10⁹/L carries cumulative risk of intracranial hemorrhage, gastrointestinal bleeding, and other life-threatening events. Long-standing active SLE simultaneously drives irreversible organ damage — lupus nephritis, neuropsychiatric involvement, cardiovascular complications — that narrows the treatment window further.
T-cell fitness degrades with each salvage attempt. CAR-T manufacturing depends on collecting T-cells that are functional enough to be engineered and expanded. Repeated rounds of immunosuppressive therapy exhaust the native T-cell compartment. The longer a patient waits, the higher the probability that their own cells will be too depleted or dysfunctional for successful autologous CAR-T production. At that point, the only remaining option may be allogeneic (donor-derived) CAR-T, which introduces additional complexity.
Lymphodepletion conditioning becomes riskier. Patients who progress to end-stage organ failure tolerate the pre-infusion conditioning regimen poorly. Metabolic complications during the "vein-to-vein" period increase, and the margin for managing CRS or other infusion-related events shrinks.
The clinical logic is straightforward: the earlier a refractory patient is evaluated for CAR-T eligibility, the better the starting material and the wider the safety margin.
Why Beijing: Volume, Integration, and Rapid Innovation
Two features distinguish Beijing's top centers from most international CAR-T programs for autoimmune indications.
First, sheer clinical volume. China leads the world in registered CAR-T clinical trials (1,006 vs. 549 in the US, per ClinicalTrials.gov). Beijing's specialized hematology units treat CAR-T patients at a pace that builds pattern recognition for side-effect management — particularly CRS grading and low-dose tocilizumab protocols that preserve CAR-T expansion while controlling toxicity. A dedicated CAR-T unit in Beijing may manage more CRS cases in a single week than many European regional centers handle in a quarter.
Second, dual-discipline integration. SLE-ITP sits at the intersection of rheumatology and hematology. In many Western systems, these are separate departments at separate institutions, requiring cross-referral and coordination delays. At Peking Union Medical College Hospital, the nation's #1-ranked rheumatology department and a hematology division with direct CAR-T capability operate on the same campus. The team that published the NEJM case had immediate access to both disciplines without referral friction.
Peking Union Medical College Hospital (PUMCH)
Founded in 1921 by the Rockefeller Foundation. China's premier National Medical Center and the designated national referral center for complex and critical diseases. Its Rheumatology & Immunology department has been ranked #1 nationally for 14 consecutive years. PUMCH pioneered the world's first CD19 CAR-T therapy for SLE-ITP. The hospital operates four campuses with 2,000+ beds, 29 national key clinical specialties, and dedicated CAR-T infusion units with laminar flow hematology wards. Total hospital outpatient volume exceeded 4.03 million visits in 2023; hospital-wide surgical volume reached 73,448 in the same year. The rheumatology department maintains 68 dedicated beds.
Source: Peking Union Medical College Hospital Official Report
Peking University Institute of Hematology
Founded in 1981 by Academician Lu Daopei. Asia's largest and one of the world's top-five hematopoietic stem cell transplant centers. Designated as China's first National Clinical Research Center for Hematologic Diseases in 2019. The institute pioneered the globally recognized "Beijing Protocol" for haploidentical transplantation, achieving 70%+ long-term disease-free survival in acute and chronic leukemia. Its cellular therapy platform treated 115+ B-ALL patients with CD19 CAR-T therapy between 2017 and 2021 and leads multi-target CAR-T research for lymphoma and myeloma. Annual outpatient volume exceeds 140,000. Annual HSCT case volume is approximately 800. The institute operates 370+ beds, including 80+ laminar flow beds.
Source: Peking University Institute of Hematology Official Report
The Specialists Leading This Work
Dr. Z — PUMCH Hematology Director of the Hematology Teaching & Research Division. Chief Physician, Professor, PhD Supervisor. Over 35 years in hematology; former Director of the Hematology Department at PUMCH. Completed postdoctoral training at UCLA (1998–2000). Pioneered autologous HSCT for POEMS syndrome in China. Led the world's first CAR-T therapy for SLE-ITP, published in the New England Journal of Medicine (2024). Over 50 publications in journals including Blood and Leukemia.
Source: Peking Union Medical College Hospital Official Description
Dr. W — Peking University Institute of Hematology / Genome Editing Research Professor of Life Sciences. Director of Peking University Genome Editing Research Center. Principal Investigator at BIOPIC and ICG. Over 20 years of experience. Completed postdoctoral training at Stanford University (1999–2004) under Nobel Laureate Stanley Cohen. PhD in Genetics from Michigan State University (1995–1999). Published in Cell (2025), Nature, Nature Biotechnology, PNAS, and Molecular Cell. Principal Investigator for the SPPL3 universal CAR-T breakthrough featured in Cell (August 2025). Founder and Scientific Advisor for EdiGene Inc.
Source: Peking University School of Life Sciences Official Description
If your case involves refractory autoimmune cytopenia and you're unsure whether your current records — lab trends, bone marrow reports, treatment history, or imaging — are sufficient for an eligibility review, you can email what you already have to contact@pandamedglobal.com. Old records are often enough to start. The team can advise on what additional documentation, if any, would be needed before a specialist review.
The Checklist
To determine CAR-T eligibility for refractory SLE-ITP, the following records are most useful. You do not need brand-new tests — existing records are often enough to start an initial review.
Autoimmune disease documentation: Recent rheumatology reports (ideally within 14 days, but older reports are still useful) confirming SLE diagnosis, disease activity indices (SLEDAI), organ involvement assessment, and current treatment regimen.
Hematologic parameters: Complete blood counts with differential, platelet count trends over time, coagulation profiles, and bone marrow biopsy reports if available.
Full treatment history: Complete list of all previous immunosuppressive therapies — corticosteroids, hydroxychloroquine, cyclophosphamide, mycophenolate, tacrolimus, cyclosporine, rituximab, belimumab, and others — with approximate dates and response assessment for each.
Organ function tests: Recent renal function (creatinine, eGFR), liver function panels, cardiac evaluation (echocardiogram with LVEF, ideally >50%), and pulmonary function testing to assess fitness for lymphodepletion conditioning.
Infectious screening: HIV, HBV, HCV, CMV, and EBV serology; QuantiFERON-TB Gold test.
If you're missing some of these, send what you have. The initial review can often proceed with partial records, and the team will tell you exactly what else is needed.
Family & Logistics
CAR-T therapy requires an extended stay in Beijing — typically several weeks for conditioning, infusion, and the critical post-infusion monitoring period. For international patients traveling with family members, questions about medical visas, sterilized long-term housing near hospital districts, appointment coordination, professional medical translation, and on-the-ground nursing support during monitoring are all common and practical.
For logistics guidance or to ask what to prepare before traveling, email contact@pandamedglobal.com.
About the Author
Ryan Li is a Beijing-based medical concierge and the founder of PandaMed. With a strict compliance background, he helps international patients navigate China's top public hospitals safely. He secures direct access to Chief Physicians for complex hematological and autoimmune cases.
Medical Disclaimer
This information is for educational purposes only and is not medical advice. PandaMed is a care coordination service, not a medical provider. Always consult a qualified hematologist or rheumatologist for your specific condition.
References
New England Journal of Medicine (2024) — First reported CD19 CAR-T therapy for SLE-ITP
Peking Union Medical College Hospital Official Report — Hospital volume, department data, and clinical outcomes
Peking University Institute of Hematology Official Report — Institute volume, HSCT data, and CAR-T clinical data
Peking Union Medical College Hospital Official Description — Specialist profile (Dr. Z)
Peking University School of Life Sciences Official Description — Specialist profile (Dr. W)
ClinicalTrials.gov — Global CAR-T clinical trial registration data
2026 Cost Analysis, Sylk Health — Comparative cost data