Sequential Multi-Target CAR-T for Relapsed/Refractory DLBCL in Beijing Hospitals
When diffuse large B-cell lymphoma comes back after multiple lines of therapy, the next decision is rarely simple. Many patients are no longer comparing routine options, but trying to find out whether there is still a credible path forward after transplant failure, prior CAR-T, or central nervous system involvement. That is where Beijing’s top hematology centers become relevant, especially for international patients looking at high-volume cellular immunotherapy programs with experience in technically difficult salvage cases.
Quick Decision Snapshot
| Decision Point | US/EU Typical Pathway | Top Beijing Public / Specialized Hematology Centers |
|---|---|---|
| Specialist wait time | 5 to 10 weeks | 4 to 8 weeks |
| Multidisciplinary review | Access usually runs through tertiary lymphoma or transplant referral pathways, often with limited slots for post-transplant relapse and prior CAR-T failure. | High-volume hematology centers routinely review complex cases involving prior autologous transplant, allogeneic transplant, donor chimerism, antigen profiling, and sequential CAR-T planning. |
| Out-of-pocket cost | $488,000 to $760,000 for commercial CAR-T | $50,000 to $80,000 in the user-provided comparison |
If you want to know whether your current pathology, PET-CT, marrow report, or prior CAR-T records are enough for an initial review, you can send existing records to contact@pandamedglobal.com. Old records are often enough for an initial records check, and you can also ask what to prepare next.
Why These DLBCL Cases Are Different From Routine Relapse
The cases in this draft are not typical salvage lymphoma scenarios. They involve patients who already failed treatments that many centers would consider major escalations.
One reported patient was a 38-year-old woman with DLBCL, ABC subtype, diagnosed in 2018. She relapsed after R-CHOP, autologous transplant, autologous CD19 CAR-T, and allogeneic transplant. By the time of relapse in 2020, the disease had already escaped multiple intensive interventions, including treatment given in the setting of established donor chimerism. That makes the case unusually difficult because post-allogeneic relapse is rare, and it raises immediate questions about immune escape, donor-derived strategies, and graft-versus-host disease risk.
Another was a 29-year-old man with stage IV DLBCL, MCD genotype, diagnosed in 2020. He failed R-CHOP × 6, R-DHAP, R-MA, and autologous hematopoietic stem cell transplant plus three cycles of dual-target CAR-T, then relapsed in 2022. His disease involved widespread extranodal sites, including the CNS, bone, and liver. This is the kind of biologically aggressive, heavily pretreated lymphoma many clinicians would regard as exceptionally hard to control.
The third patient was a middle-aged woman with triple-refractory DLBCL and CNS involvement, diagnosed in 2016, who continued to progress after R-CHOP and multiple salvage regimens. In practical terms, this is the sort of disease course where standard single-step planning often breaks down. The question is no longer whether the patient has had “enough” therapy, but whether the next therapy can still match the biology of the relapse.
Why Sequential Multi-Target CAR-T Has Drawn Attention
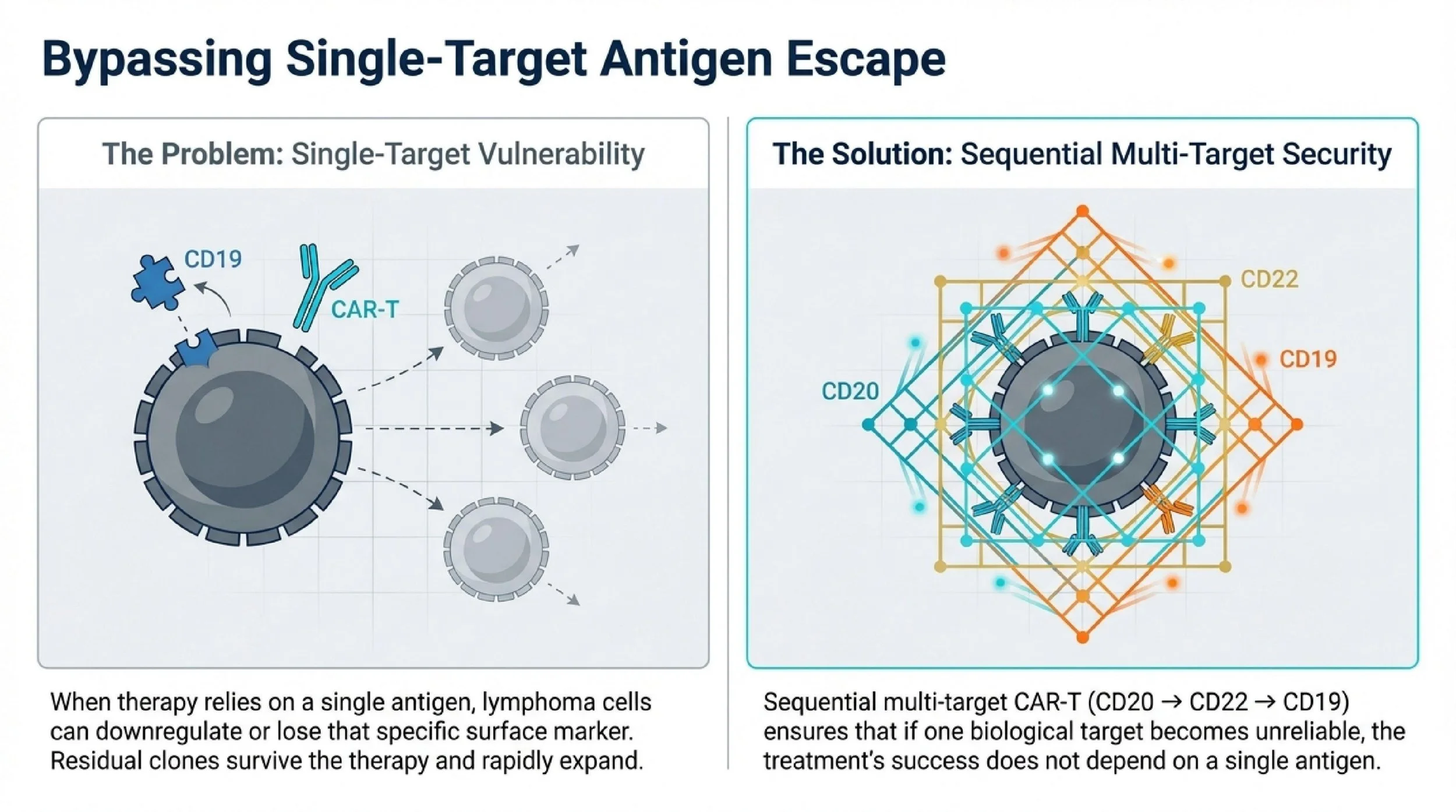
The central idea in the Chinese strategy described here is straightforward: single-target CAR-T can fail because the tumor changes the target. In relapsed/refractory DLBCL, one major resistance mechanism is antigen escape, meaning lymphoma cells reduce or lose expression of the antigen being attacked.
That is the rationale behind sequential multi-target CAR-T using CD20, CD22, and CD19. Instead of relying on only one antigen, the treatment sequence is designed to keep pressure on the lymphoma across multiple target expressions. For a patient who has already failed prior CAR-T or who has a biologically unstable relapse, this is not a small technical adjustment. It changes the salvage logic.
The reported clinical highlights are notable. In the post-transplant relapse setting, this strategy was associated with 4-year sustained complete remission in what is described as the world’s first reported case of relapsed/refractory DLBCL after allogeneic hematopoietic stem cell transplant achieving long-term durable complete remission through sequential infusion of haploidentical CD20/CD22/CD19 CAR-T cells. According to the case description, it also provided the first proof that donor-derived haploidentical CAR-T could produce long-term remission after allogeneic transplant relapse without severe GVHD.
Additional outcomes in the user’s source set include a 71.4% sustained remission rate for sequential dual-target CD19/CD20 CAR-T at a median follow-up of 24.7 months. A triple-target program in Burkitt lymphoma showed 100% remission, with an 18-month complete remission rate of 86.9%. Burkitt lymphoma is not the same disease as DLBCL, so these data should not be treated as interchangeable. Still, they help illustrate the development of multi-target CAR-T platforms in Beijing’s cellular therapy environment.
The safety profile cited here is also important: grade 3 or higher CRS in 9.5%, ICANS in 0%, and no treatment-related deaths.
When Waiting Can Reduce the Next Good Option
For aggressive relapsed DLBCL, waiting is not always a neutral choice. That is especially true after multiple lines of therapy, prior CAR-T exposure, or known CNS-risk disease.
The first issue is antigen evolution. Every relapse can increase the chance that the tumor will no longer express CD19 or CD20 in a stable way. If that happens, a later CAR-T strategy may become less effective or more limited. This is why repeat antigen assessment can matter so much in refractory disease.
The second issue is T-cell quality and manufacturing feasibility. Heavily pretreated patients may have more exhausted T cells, especially after repeated chemotherapy used just to hold the disease in place. If a patient is still being considered for autologous CAR-T, this matters directly. If the treatment plan involves donor-derived or transplant-background strategies, timing still matters because the patient’s overall condition and disease burden can shift quickly.
The third issue is extramedullary disease and CNS spread. In the cases summarized here, one patient had extensive extranodal disease involving the CNS, bone, and liver, and another had refractory disease with CNS involvement. Once aggressive lymphoma becomes more anatomically dispersed, salvage becomes more difficult. The longer the disease stays uncontrolled, the narrower the therapeutic window may become.
The user also cited Journal of Clinical Oncology 2022 on disease burden and outcomes after commercial tisagenlecleucel in pediatric and young adult B-cell acute lymphoblastic leukemia. That paper is not a DLBCL study, so it should not be overextended. But the broader principle is still useful: higher disease burden can make cellular therapy outcomes worse and care more complicated.
If the case is complex, or you are unsure whether MRI, pathology, transplant records, infusion summaries, or older reports are enough, email what you already have to contact@pandamedglobal.com for record-preparation and next-step guidance. Existing records are often enough to start.
Why Beijing’s High-Volume Centers Matter in This Niche
Not every hospital that offers CAR-T is equally experienced with the same type of case. For international patients with relapsed/refractory DLBCL, especially after prior CAR-T or transplant, the more relevant question is whether the center routinely handles this exact failure pattern.
According to the user-provided data, Beijing L Hospital recorded 2,782 CAR-T clinical trial enrollments and 1,662 hematopoietic stem cell transplants in 2025, with 230 beds. It is part of the Lu Daopei Medical Group, founded by Academician Lu Daopei, and is recognized for blood disease diagnosis and treatment. The hospital description includes advanced diagnostic platforms such as ZEISS microscopy systems, Cytek full-spectrum flow cytometers, BD Canto II and Aria cell sorters, and GE Revolution HD CT scanners.
Beijing B Hospital is described as a specialized hematology-focused hospital under the GoBroad Medical Group, with 170 beds, 200+ annual pediatric CAR-T infusions, and 1,000+ total hematopoietic stem cell transplants in the user’s summary table. Its broader hospital profile notes Siemens 64-slice spiral CT, Siemens 3.0T MRI, and a 5,000-square-meter laboratory platform covering cell morphology, flow cytometry, chromosome and FISH analysis, and genetic testing. The hospital is also described as a leading site in the development of sequential multi-target CAR-T therapies involving CD19, CD20, and CD22, with related publications in Frontiers in Immunology, Blood, and Cytotherapy.
The volume comparison in the draft is striking. The user cites 30 to 50 annual CAR-T cases as a typical Western range versus 200+ annual cases in Beijing, along with global clinical trial counts of 549 versus 1,006. Even when care models differ across countries, the underlying point is clinically meaningful: in cellular therapy, repeated exposure to complex toxicity management and relapse biology can shape how a center handles CRS, ICANS surveillance, bridging decisions, donor-background cases, and infusion timing.
Hospital Navigation Is Often Harder Than Patients Expect
One practical problem for international patients is that top Chinese hematology teams usually do not manage direct English-language inquiries in the way many overseas families expect. Another is that public hospital registration systems can be difficult for foreigners, especially when specialty appointments sell out early or require local documentation.
That matters more in CAR-T than in many other service lines because complex cases are often screened only after a full medical record pre-review. A patient with prior transplant, CNS disease, or earlier CAR-T exposure is not simply booking a routine outpatient slot. The team usually needs to review pathology, treatment sequence, antigen expression, transplant background, and recent imaging before deciding whether there is a feasible pathway.
Senior Specialist Profiles
For this article, anonymous profiles are more appropriate than full academic biographies.
Dr. L is a senior hematology-oncology leader at Beijing Hospital with 30+ years of experience and a major role in advanced blood cancer care and cellular therapy programs.
Dr. T is a senior hematology and immunotherapy specialist at Beijing with 40+ years of experience and leadership experience in 4,000+ CAR-T cases according to the hospital source material.
In highly refractory DLBCL, this level of experience matters most when the disease has already moved beyond standard treatment logic.
The Checklist
You do not need brand-new records to begin. Existing records are often enough to start. The most useful materials for relapsed/refractory DLBCL review usually include the following:
Bone marrow biopsy and flow cytometry reports, ideally including CD19, CD20, and CD22 expression
Pathology report and immunohistochemistry from diagnosis and any relapse biopsy
Genetic or NGS reports, especially if they include MYD88, CD79B, TP53, BCL2, and MYC
Full treatment timeline with dates for R-CHOP, salvage regimens, transplant, prior CAR-T, and best response to each line
PET-CT reports with Deauville score when available
Brain MRI reports if there is known or suspected CNS involvement
Recent cardiac and renal function data, including LVEF and creatinine clearance if available
Prior transplant records, including donor chimerism, GVHD history, and current or prior immunosuppression
Hospital discharge summaries and prior CAR-T infusion records
Any translations you already have, although original-language records can still be useful for an initial check
Family / Logistics
Families who need help understanding visas, lodging, appointments, translation, family travel, or broader Beijing care coordination can email contact@pandamedglobal.com for practical guidance on next steps.
Author Bio
Ryan Lee is a Beijing-based medical concierge and the founder of PandaMed. With a strict compliance background, he helps international patients navigate China's top public hospitals safely. He secures direct access to Chief Physicians for complex cases.
Medical Disclaimer
This information is for educational purposes only and is not medical advice. PandaMed is a care coordination service, not a medical provider. Always consult a qualified doctor for your specific condition.
References
Chin Med J (Engl). 2019. A retrospective analysis of real-world outcomes of elderly Chinese patients with diffuse large B-cell lymphoma
Adv Ther. 2022. Tafasitamab Plus Lenalidomide Versus 3 Rituximab-Based Treatments for Non-Transplant Eligible Relapsed/Refractory Diffuse Large B-Cell Lymphoma: A Matching-Adjusted Indirect Comparison
Biomark Res. 2026. A global multidimensional analysis of the chimeric antigen receptor T-cell therapy clinical trial landscape and development trends
2026 Cost Analysis of Sylk Health
Journal of Clinical Oncology. 2022. Disease Burden Affects Outcomes in Pediatric and Young Adult B-Cell Lymphoblastic Leukemia After Commercial Tisagenlecleucel